Overview
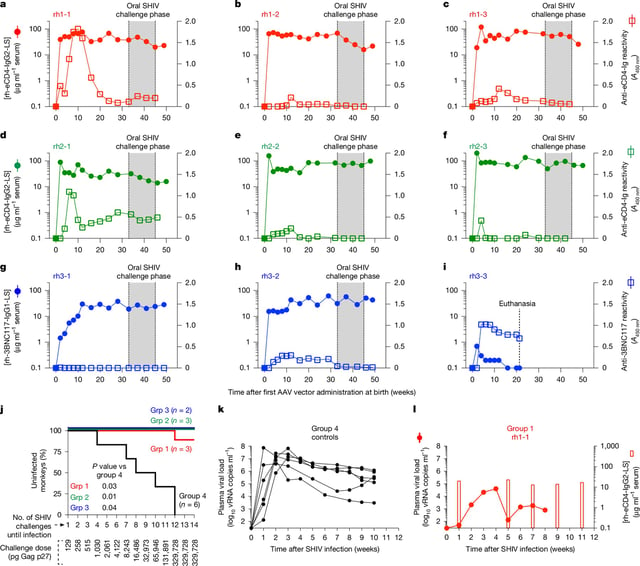
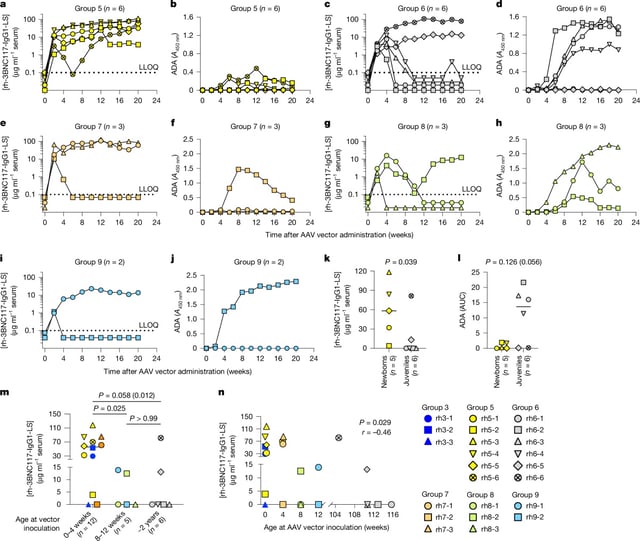
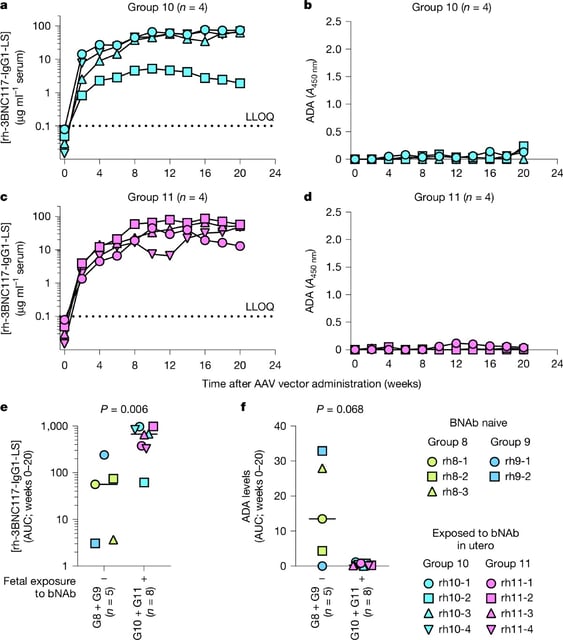
- A Nature study found that rhesus macaques given a single AAV-based broadly neutralizing antibody injection at birth remained protected from HIV for at least three years without boosters.
- The approach exploits a naturally tolerant neonatal immune window to prevent anti-drug responses that undermine gene therapies in older infants.
- AAV vectors deliver antibody genes to muscle cells, turning them into long-lived factories that continuously produce broadly neutralizing anti-HIV antibodies.
- Researchers are preparing Phase 1/2 newborn safety trials, focusing on vector immunogenicity, coverage against diverse HIV strains and scalable manufacturing methods.
- If translated successfully, the one-time injection could dramatically reduce mother-to-child HIV transmission during breastfeeding in high-risk regions.