Overview
- The randomized multicenter Phase II LUBI-CKD trial across nine Japanese centers enrolled 150 patients with moderate chronic kidney disease.
- Lubiprostone slowed the decline in estimated glomerular filtration rate versus placebo in a dose-dependent pattern.
- Participants receiving 8 µg or 16 µg doses had slower loss of kidney function than those on placebo.
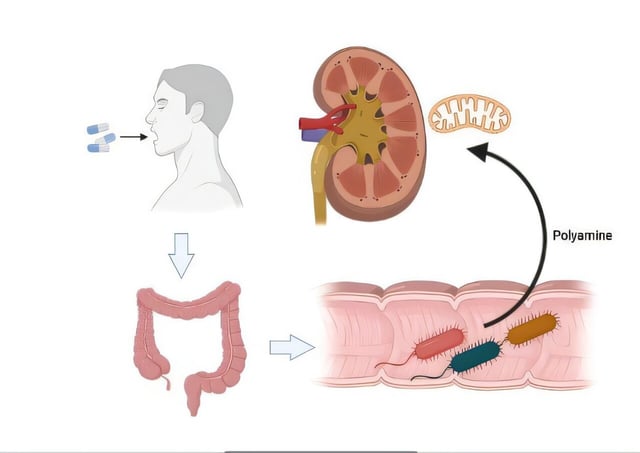
- Mechanistic analyses tied gut microbiome changes to higher spermidine levels, improved mitochondrial function, and reduced inflammation.
- The peer-reviewed report appears in Science Advances, and investigators are preparing a Phase III trial and biomarker studies; the primary endpoint was eGFR slope rather than clinical events.